Table of Contents
List of items eligible for Continued Dispensing has been expanded to cover all the essential drugs
Full PBS quantity
PBS price
No repeats
Other Continued Dispensing rules still apply (i.e. once per 12 months period)
Emergency Dispensing #
Non-PBS Price
PBS Quantity or 1 pack
No repeats
Other Emergency Dispensing rules still apply (this depends on which state they are in)
Generally, for PBS-eligible medication, use Continued Dispensing first as preference, to allow PBS Pricing.
If item is Private or not-PBS eligible for Continued Dispensing, use the Emergency Dispensing Provisions.
LOTS Streamlined Workflow
LOTS Classic Workflow
AWD
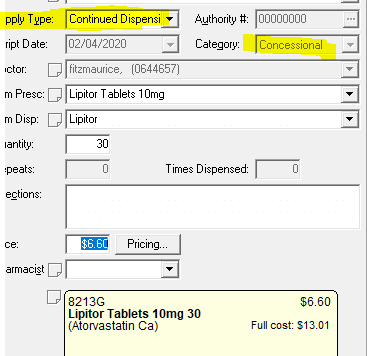
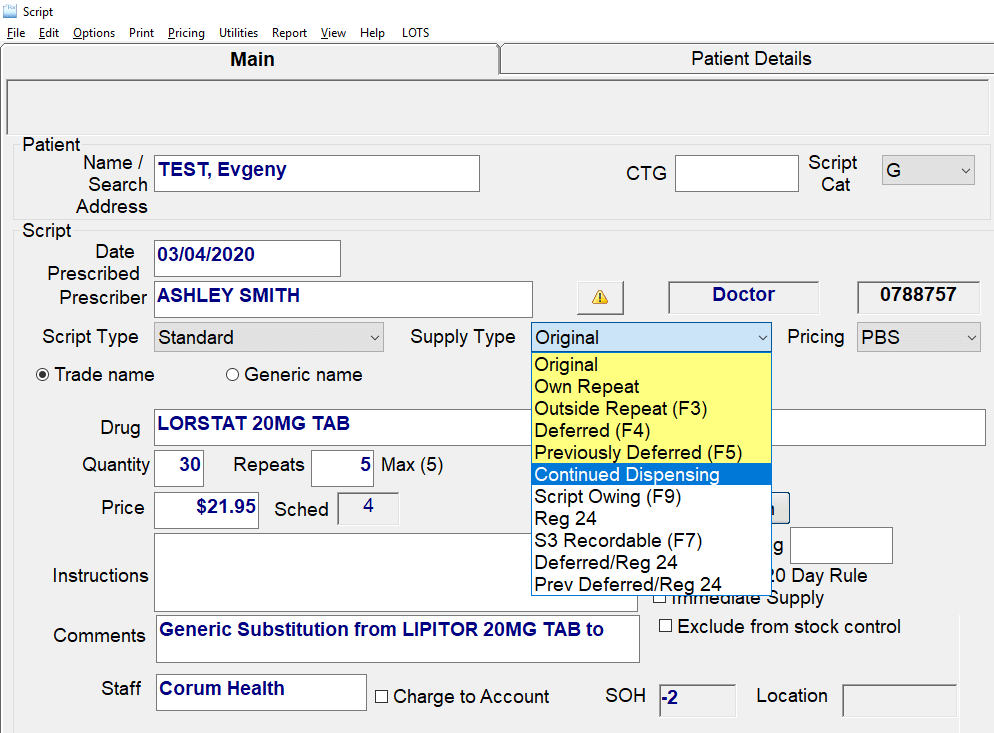
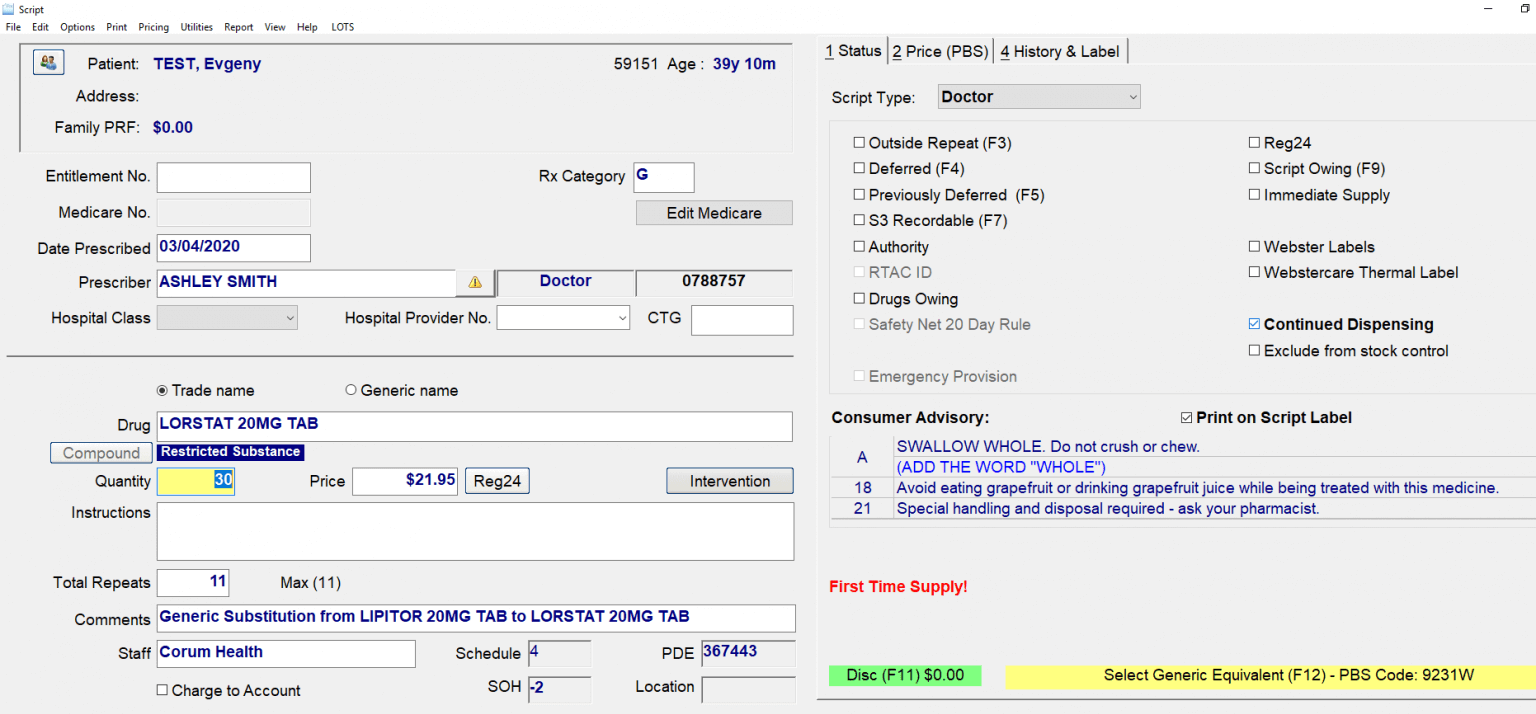
CCD Continued Dispense
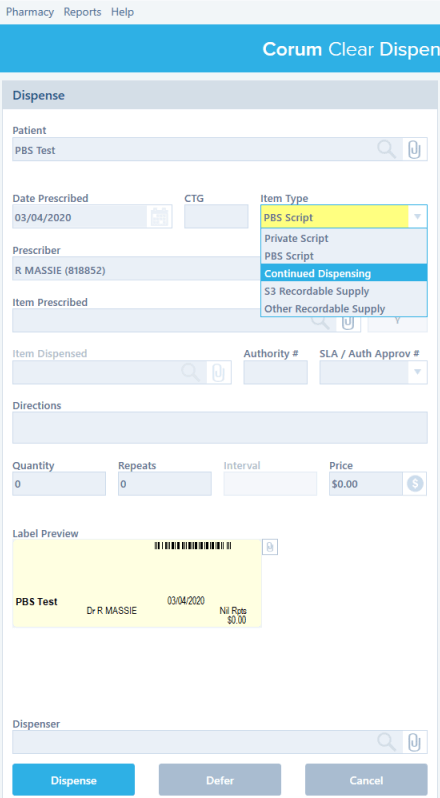
CCD Other Recordable Supply
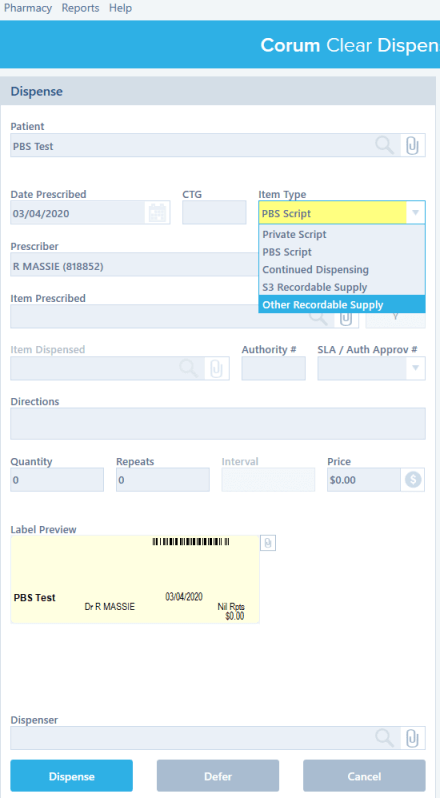
Continued Dispensing #
Continued Dispensing is an option available to pharmacies to dispense medicines that are immediately needed by the patients without having to first provide a prescription. This minimizes treatment interruption due to a patient’s inability to obtain a timely prescription renewal.
Eligibility
The following eligibility criteria must be met to dispense using Continued Dispensing:
- For Suppliers: Only community pharmacy and approved suppliers under Section 90 and Section 90A
- For Patients: Only a patient who is an Australian resident with a current Medicare card or with a Reciprocal Health Care Agreement.
-
For Medicines: Only eligible medicines, including medication brand which are outlined in the National Health (Continued Dispensing) Determination 2012 National Health Act 1953. Some of these medicines include:
- Oral Hormonal Contraceptives
- Lipid Modifying Agents (HMG CoA reductase inhibitors)
Note: To avail PBS or RPBS subsidy, only the medicines that are listed in the schedule of Pharmaceutical Benefits can be availed.
Configuring Continued Dispensing #
LOTS filters eligible medications, only if the Continued Dispensing option is registered and enabled in LOTS for your pharmacy. To do so, you need to do the following:
While dispensing a script, you can record medicines being supplied under continued dispensing from LOTS and generate the following forms:
- Modified Repeat Authorisation Form: This form is used to capture the details of the Continued Dispensing supply and for the patient, or their agent, to sign and confirm that they have received the medicine.
- Consumer Declaration and Prescriber Notification Form: This form is used to
- Demonstrate that the patient, or their agent, understands that the supply has been undertaken without consultation from their prescriber.
- Notify the patient’s prescriber, that a continued dispensing supply has been given.
- Notify the most recent prescriber in writing, within 24 hours that the medicine has been supplied.




